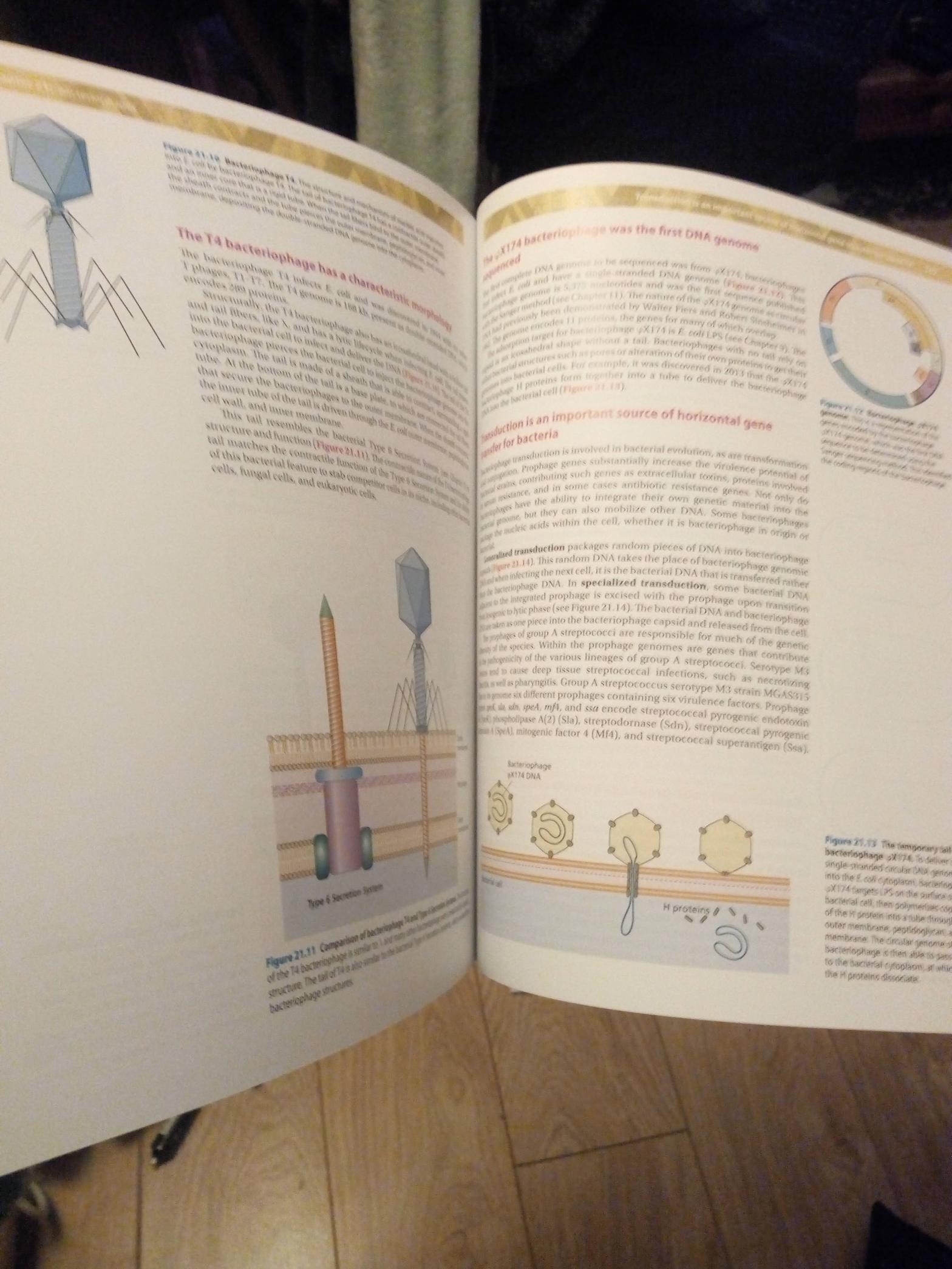
Bacterial Genetics and Genomics book Discussion Topic: Chapter 20, question 14
This is the first time since I started this blog that I have been able to do it with a physical copy of the book at hand! It is so exciting to finally have tangible evidence of my hours of writing and to see the excellent finished product made possible through Patrick Lane’s talents bringing my pencil drawings to life in full-color illustrations and designing the cover, as well as the production team putting on the final touches.
It seems that most of the topics in the news and in academic circles on Twitter are in some way related to COVID-19. With that in mind, I have chosen to blog about azithromycin, which has been used in the treatment of some COVID-19 patients. Azithromycin is an antibiotic, which has antibacterial activity against some species of disease-causing bacteria. It is not used as an antibacterial against Pseudomonas aeruginosa, but azithromycin does have antibiofilm activity. Biofilms can be formed by some bacterial species like P. aeruginosa on natural and artificial surfaces and are a major health problem, due to their resistance to treatment. Biofilms are explored in Bacterial Genetics and Genomics in Chapters 5, 6, and 13.
To explore new research in the application of azithromycin for antibiofilm treatment of P. aeruginosa, I have selected a January 2020 paper that proposes antibiotic delivery directly to the biofilm location.
Lim DJ, Skinner D, Mclemore J, Rivers N, Elder JB, Allen M, Koch C, West J, Zhang S, Thompson HM, McCormick JP, Grayson JW, Cho DY, Woodworth BA. In-vitro evaluation of a ciprofloxacin and azithromycin sinus stent for Pseudomonas aeruginosa biofilms. Int Forum Allergy Rhinol. 2020 10(1):121-127. doi: 10.1002/alr.22475.
Rather than using a systemic antibiotic, a targeted delivery approach will mean that the antibiotic is at the concentration needed at the site of infection so that it can most effectively eliminate the bacteria and also possibly reducing side effects. In this case, the concern is P. aeruginosa chronic rhinosinusitis (sinus infection). Biofilms form in the sinuses and are very difficult to treat effectively using oral or IV antibiotics. Instead, these authors have recommended making a sinus stent that will deliver antibiotics directly to the biofilm. The ciprofloxacin-azithromycin sinus stent used in this study was constructed with two antibiotics, which both modulates the release of the drugs due to their different hydrophobic properties and also gives the stent two different antibacterials to combat the biofilms. Although we tend to study single species biofilms in the lab, those that occur in nature – and inside us – are often complex communities, so a multi-drug approach may be a good idea.
Coating the stents with both antibiotics prolonged the release of the ciprofloxacin, versus stents that were just coated with ciprofloxacin on its own. This is due to the hydrophobic nature of azithromycin, which was coated on the outside of the stents and meant that the ciprofloxacin wasn’t released in just one burst, as it was when there was no azithromycin.
In vitro, the stents with azithromycin and ciprofloxacin had good activity against P. aeruginosa biofilms. Interestingly, the authors note that they included azithromycin in the design of the stent only because of its hydrophobic properties, so that it would stop the burst release of the ciprofloxacin. They mention that it has positive anti-inflammatory benefits in treatment and that they will look at increasing concentrations of both antibiotics when moving to in vivo studies, but there is no mention of the antibiofilm activity of azithromycin against P. aeruginosa, which is known and used as a positive control for experiments investigating other antibiofilm drugs. This is another positive in favor of the decision of Lim et al. to use azithromycin as the hydrophobic outer drug for their sinus stent.
Tateda K, Comte R, Pechere JC, Köhler T, Yamaguchi K, Van Delden C. Azithromycin inhibits quorum sensing in Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2001 45(6):1930-1933.
Skindersoe ME, Alhede M, Phipps R, Yang L, Jensen PO, Rasmussen TB, et al. Effects of antibiotics on quorum sensing in Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2008 52:3648–3663. doi: 10.1128/AAC.01230-07.
Seleem NM, Abd El Latif HK, Shaldam MA, El-Ganiny A. Drugs with new lease of life as quorum sensing inhibitors: for combating MDR Acinetobacter baumannii infections. Eur J Clin Microbiol Infect Dis. 2020. doi: 10.1007/s10096-020-03882-z.
Repurposing of antibiotics and thinking about them in new ways in which we can use them to fight infections are important points for discussion. Understanding bacterial genetics and genomics gives us insights into biofilms and antimicrobial susceptibilities that can drive ongoing and future research.
In case you haven’t got a copy yet, there is still a sale going on for the book (20% off) and ebook (35% off): https://www.routledge.com/Bacterial-Genetics-and-Genomics/Snyder/p/book/9780815345695