Bacterial Genetics and Genomics book Discussion Topic: Chapter 2, question 14
It has long been a defining difference that bacterial cells, like E. coli, have coupled transcription-translation and eukaryotic cells, like animals and plants, make their mRNA in the nucleus and their proteins in the cytoplasm. This is mostly the case. Mammals did break the rules and got found out a few years ago (see Coupled Transcription and Translation within Nuclei of Mammalian Cells). But, it seemed clear that the proteins made in the mammalian nucleus weren’t the main process for these cells and that bacterial cells were the clear leaders in doing coupled transcription-translation.
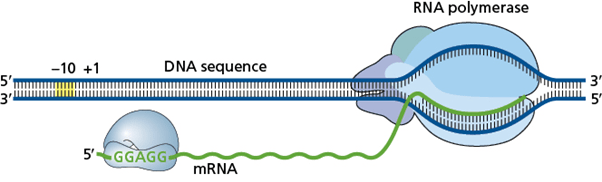
The transcription-translation complex (TTC) is the physical association of the RNA polymerase (RNAP), which is transcribing the DNA into mRNA, and the ribosome (containing rRNA and ribosomal proteins), which is translating mRNA into protein with the help of tRNAs carrying amino acids. The TTC is also known as the expressome.
It’s been discovered that bacteria have been breaking the rules as well. As we often find, when we try to define our world, we will come across exceptions to our rules and classifications, and we need to be able to reappraise our understanding as we learn more and do more research. This is one such example. Coupled transcription-translation is an ideal example from microbiology. A lot of what we know in microbiology, especially insights gained in early microbiology, is based on research in E. coli that was extrapolated across to other bacterial species. The more we learn about other bacterial species, the more we learn about how diverse they are and how much they do not do things the same as E. coli, like coupled transcription-translation.
In August of 2020, Grace E. Johnson, Jean-Benoît Lalanne, Michelle L. Peters, and Gene-Wei Li published research focused on the Gram-positive model species Bacillus subtilis that challenged the coupled transcription-translation paradigm that had been established 50 years earlier in E. coli, a model Gram-negative bacterial species.

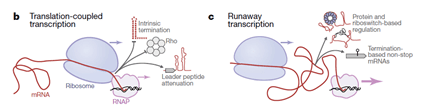
The B. subtilis RNAP was shown to transcribe DNA into mRNA much faster than the ribosomes can progress along the mRNA to conduct translation. This ‘runaway transcription’ means that the RNAP and ribosome are not coupled in transcription-translation, with RNAP moving at nearly twice the speed and leaving the translation machinery in its wake.
The differences between E. coli and B. subtilis with regards to the association between RNAP and the first ribosome conducting translation mean that there are also other differences. B. subtilis tends to rely less on Rho-dependent transcriptional termination and to have a greater prevalence of riboswitches in the mRNA leader sequences. Looking at these features, the researchers were able to suggest that ‘runaway transcription’ is not limited to B. subtilis and that there should now be two models of bacterial transcription and translation: translation-coupled transcription and runaway transcription.
There is some further reading about insights into transcription – translation in bacteria, including coupled, uncoupled, and collided E. coli expressome states in a Nature Microbiology Reviews Research Highlight article.
The Johnson et al., 2020 contribution to our wider understanding of transcription and translation in bacteria establishes a second model of ‘runaway transcription’, which is a common feature of some bacterial species, rather than coupled transcription-translation. This is an important insight into bacterial genetics and genomics and a reminder of the diversity of bacterial species.
